Computational Diagnostic Algorithms
While sample preparation technologies enable the whole genome sequencing of pathogens directly from clinical samples, once the samples have been sequenced there is a critical need for robust algorithms to interpret the sequencing data.
Our computational methods take the raw genomic data as input and transform them into the diagnostic results that can serve infectious disease clinicians and clinical microbiologists.
Keynome® ID
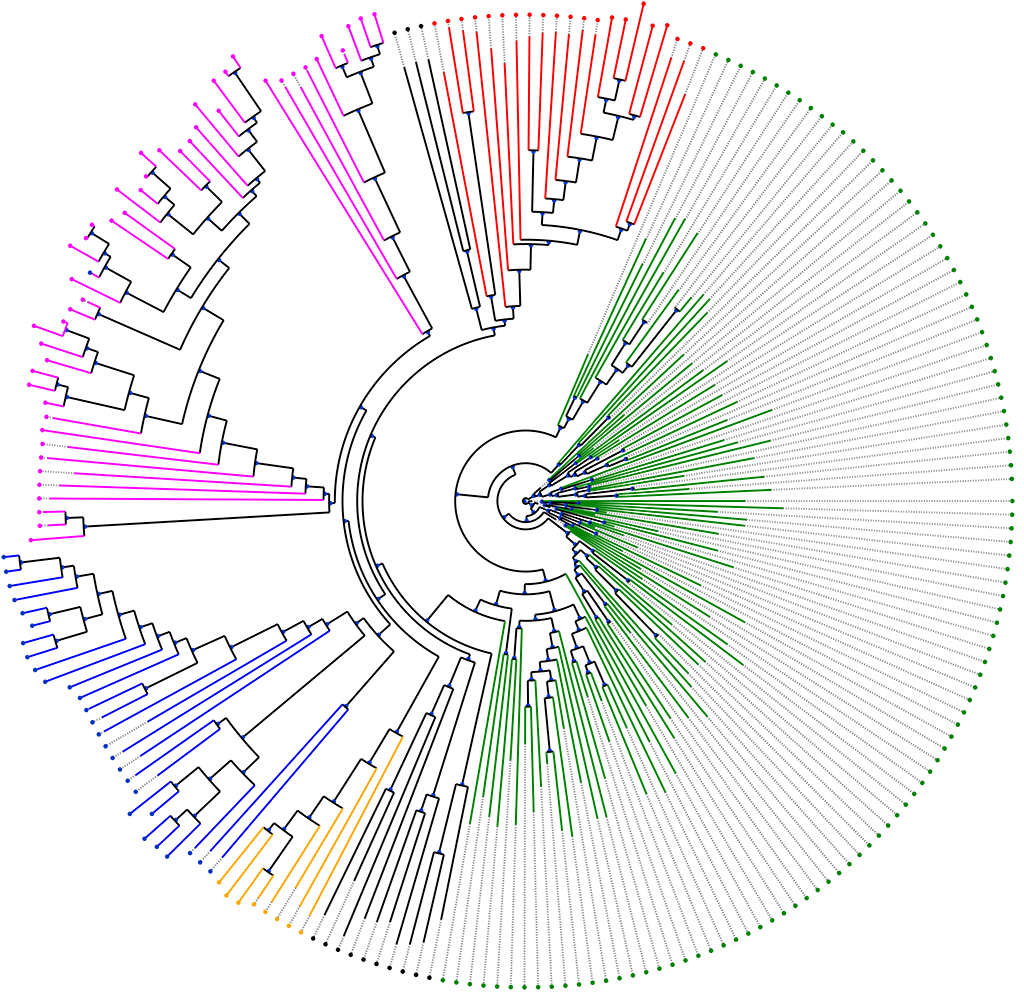
Keynome® ID is our proprietary algorithm that classifies the bacterial species present in a sample. Leveraging an internally-curated reference genome database to ensure high-accuracy across a large number pathogen species, Keynome ID provides high confidence for identifying the pathogen in clinical samples. When paired with the ultra-high enrichment of bacterial DNA using Blood2Bac preprocessing, Keynome ID determines the species of infections at concentrations as low as 1CFU/mL without being impaired by the high false-positive rates that often greatly reduce the diagnostic applicability and interpretability of hyper-sensitive molecular methods.
Keynome® gAST
Keynome® gAST is our machine learning algorithm for performing genomic Antibiotic Susceptibility Testing (gAST). Keynome gAST determines the antibiotic resistance and susceptibility profile of a pathogen directly from whole genome sequencing data. Keynome is a modern, novel machine-learning algorithm that leverages our proprietary training dataset and recent advances in the field of artificial intelligence in order to provide high accuracy gAST across a comprehensive range of species and antibiotic drugs. Unlike traditional “database look-up” approaches that identify the presence or absence of a limited set of experimentally validated resistance genes, Keynome allows for the simultaneous testing of a comprehensive set of bacterial species and resistance markers, along with the ability to identify novel resistance determinants as they emerge. Keynome gAST can be applied to the sequencing data of a clinical sample processed by Blood2Bac in order to determine the AST of a pathogen within hours of a blood draw.